¿Ha perdido la oportunidad de hacer una pregunta al Dr. Zanolli?
Apúntese a un segundo webinar el 6 de mayo de 2021 a las 13:00 CLT / 14:00 EDT
Mario Zanolli es un oftalmólogo pediátrico de Clinica Alemana de Santiago y Hospital Roberto del Río, Profesor Adjunto de la Universidad del Desarrollo. Hizo su formación en la Fundación Oftalmológica Los Andes y luego un fellow en Genética Ocular en el Hospital Wills de Philadelphia, donde fue co-autor del Manual Wills de Oculogenética.
![]() Warning: This product can expose you to chemicals including lead, which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information go to www.P65Warnings.ca.gov.
Warning: This product can expose you to chemicals including lead, which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information go to www.P65Warnings.ca.gov.
Substance Tables:
The table below lists substances which may be contained within LKC’s RETeval and RETevet products. Substances listed as Type 1 are within permissible levels in one or more of LKC’s products. Substances listed as Type 2 are used in the production of some components used in LKC products and may be present at trace levels, but are typically destroyed during processing.
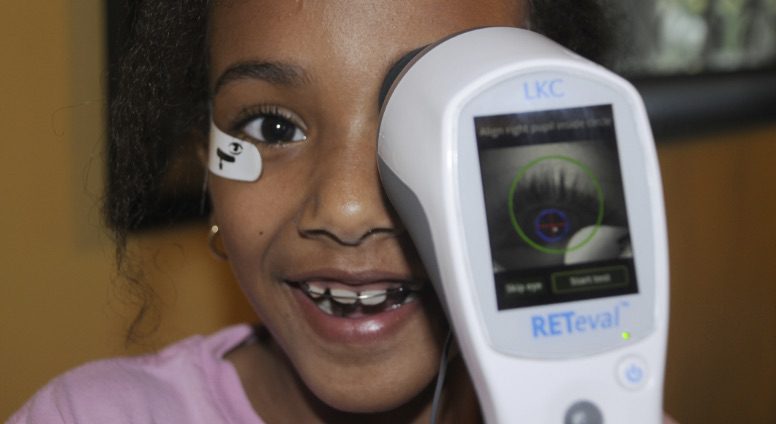
RETeval and RETevet Devices
| Substance | CAS # | Type | Listed as causing: |
| Nickel | 7440-02-0 | 1 | Cancer |
| Acrylonitrile | 107-13-1 | 2 | |
| Ethylbenzine | 100-41-4 | 2 | |
| Crystaline Silica | 14808-60-7 | 1 | |
| Lead | 7439-92-1 | 1 | Cancer Developmental Toxicity Male Reproductive Toxicity Female Reproductive Toxicity |
| Methylene Chloride | 75-09-2 | 2 | Cancer Female Reproductive Toxicity |
| Bisphenol A | 80-05-7 | 2 | |
| N-Hexane | 110-54-3 | 2 | Male Reproductive Toxicity |
